On 1 March 2017, Bangladesh, Brazil, India and South Africa, the Secretariat of the United Nations Secretary-General’s High-Level Panel on Access to Medicines and the South Centre convened a side event at the World Trade Organization (WTO) on the United Nations Secretary-General’s High Level Panel on Access to Medicines: Opportunities to Advance Health Technology and Access. This event took place on the margins of the WTO TRIPS Council.
Continue Reading →
In December 2016, Brazil submitted a proposal to the World Trade Organization entitled, ELECTRONIC COMMERCE AND COPYRIGHT (JOB/GC/113, JOB/IP/19), intended for circulation at the WTO’s General Council and the TRIPS Council. This paper will be discussed at the next session of the TRIPS Council under agenda item 13, “Work Programme on Electronic Commerce” (1-2 March 2017).
Continue Reading →
February 15, 2017, the USPTO ruled that 12 genome-editing CRISPR-Cas9 patents and one patent application assigned to the Broad Institute of Harvard and MIT did not interfere with a patent application from scientists at the University of California. A copy of the ruling is available here.
Following the ruling by the USPTO that the Broad Institute and the University of California both issued statements, as did several firms with an interest in the dispute.
On February 9, 2017, KEI filed written comments to the Office of the United States Trade Representative’s Special 301 Review process.
The Special 301 Review is an annual process carried out by the USTR to, “to identify countries that deny adequate and effective protection of intellectual property rights (IPR) or deny fair and equitable market access to U.S. persons who rely on intellectual property protection.”
Continue Reading →
In December 2016, during the twilight of President Barack Obama’s 44th presidency, the World Trade Organization (WTO) conducted a Trade Policy Review (TPR) of the United States of America. All members of the WTO are subject to review under the Trade Policy Review Mechanism (TPRM). The TPRM takes place in the “Trade Policy Review Body which is actually the WTO General Council — comprising the WTO’s full membership — operating under special rules and procedures” (Source: WTO, Trade Policy Reviews: Brief Introduction).
Continue Reading →
On the morning of Monday, 23 January 2017, India delivered the following intervention at the 140th session of the World Health Organization’s Executive Board requesting the “explicit inclusion” of the United Nations Secretary-General’s High-Level Panel Report in the “provisional agenda of the EB140 as a separate agenda item under item 8.”
In making the case for the inclusion of the UN HLP in the WHO’s Executive Board deliberations, India articulate the following:
Continue Reading →
At 11:06 P.M. on January 11, 2017, during the so-called “Vote-A-Rama”, the Senate considered an amendment put forward by Sen. Amy Klobuchar, D-Minn., and co-sponsored by Sen. Bernie Sanders, I-Vt., on the parallel importation of prescription drugs from Canada. Continue Reading →
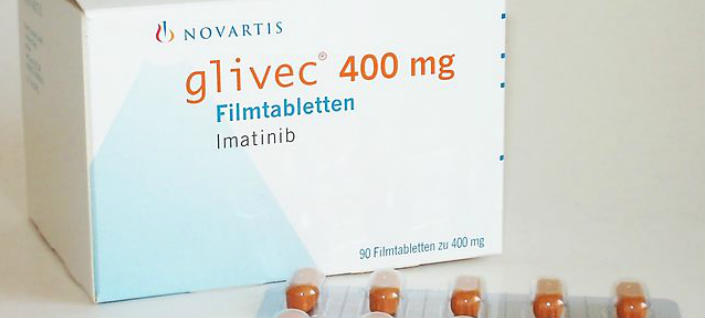
The Colombian Ministry of Health has finalized the price reduction of Glivec, Novartis’s +$47 billion leukemia drug, with a decrease in price from 368 pesos per milligram to 206.42 pesos per mg.
Continue Reading →
Matt Richtel and Andrew Pollack explore the complex policy issues involved with the pricing of products based on taxpayer-funded inventions and research in a front page New York Times story published today.
Continue Reading →